
Teva Announces New Findings Highlighting Potential of ProAir® Digihaler® (albuterol sulfate) Inhalation Powder to Address Asthma Treatment Challenges

Lipegfilgrastim is the result of a two-step enzymatic reaction. Notes:... | Download Scientific Diagram
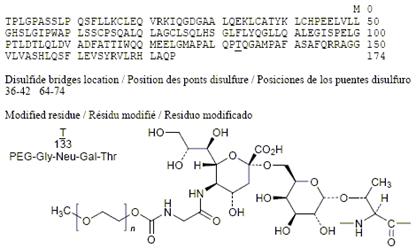
Teva Pharmaceutical has been given a green light by the European Commission (EC) for Lonquex, a rival to Amgen's blockbuster Neulasta. | New Drug Approvals

Frontiers | Pooled Analysis on the Effectiveness and Safety of Lipegfilgrastim in Patients With Urological Malignancies in the Real-World Setting | Oncology

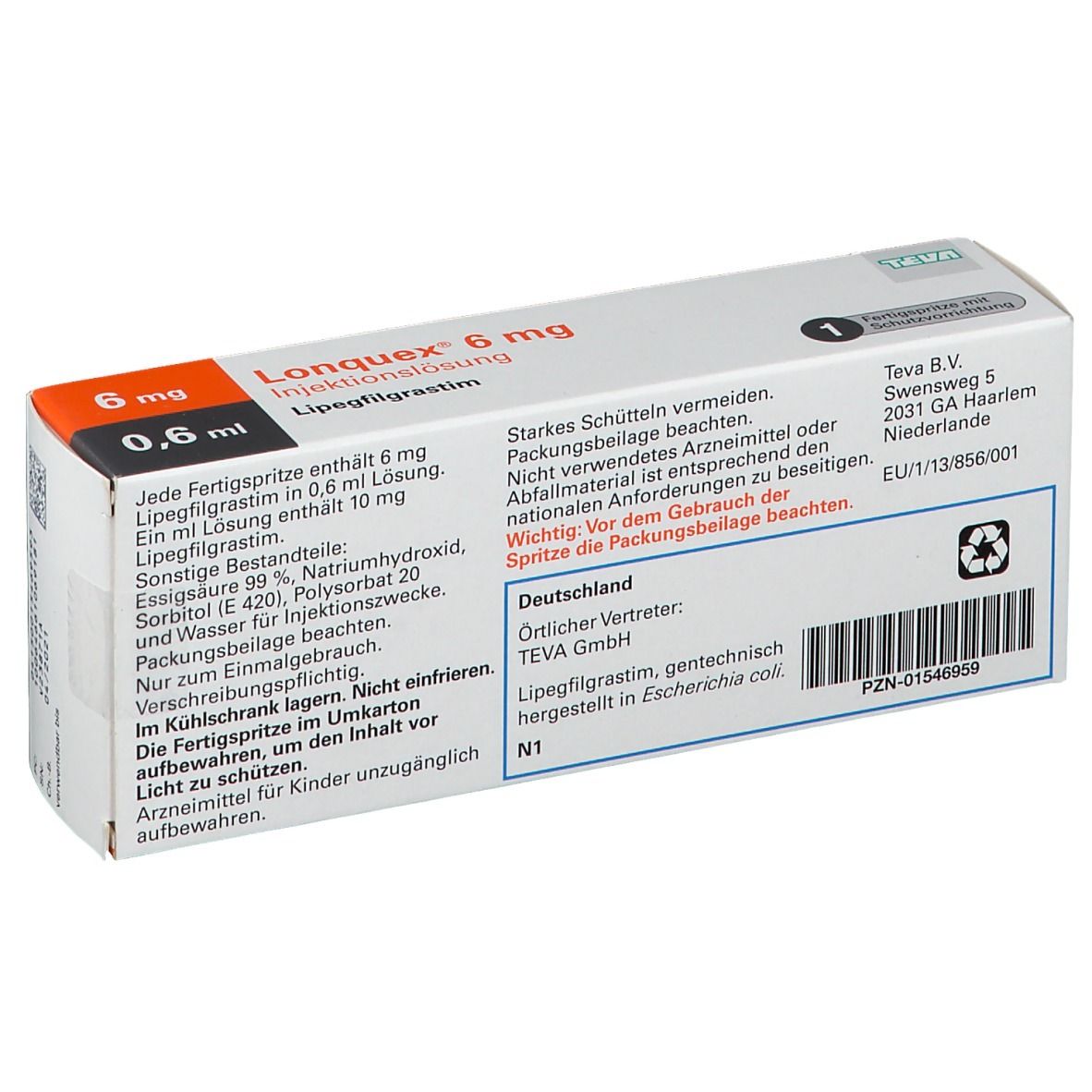
LONQUEX 10 MG/ML SOLUÇÃO INJETÁVEL COM 1 SERINGA PREENCHIDA DE 0,6 ML - LIPEGFILGRASTIM - TEVA (REFRIGERADO) - Ative Medicamentos

Neutropenia Biologic Drug Therapy International Market Report 2022: Filgrastim, Pegfilgrastim, Lenograstim, Lipegfilgrastim, Sargramostim, Antibiotic Medicine, G-CSF, Antifungal Medicine - ResearchAndMarkets.com - Times Grid
















![Buy Lonquex® [Lipegfilgrastim] Buy Lonquex® [Lipegfilgrastim]](https://pillbuys.com/25251-large_default/lonquex-lipegfilgrastim-.jpg)